Heat transfer is a branch of science in which we study about the energy transfer (in form of heat) between materials due to temperature difference. Heat is a form of energy which transfers by virtue of temperature difference.
After reading the term “Heat Transfer” one question arises in our mind that why we study it separately while we can study it in thermodynamic too.
The answer is quite simple is that in thermodynamic we study the total amount of heat transfer between two equilibrium states but in heat transfer we study the rate of heat transfer and by use of this study we can know the temperature of a material at any instantaneous time.
Example-
If we study the cooling of a body in the air then thermodynamic tell us overall heat rejected by the body for getting equilibrium state. It will not tell us how much time it takes. So heat transfer is used to answer this question.
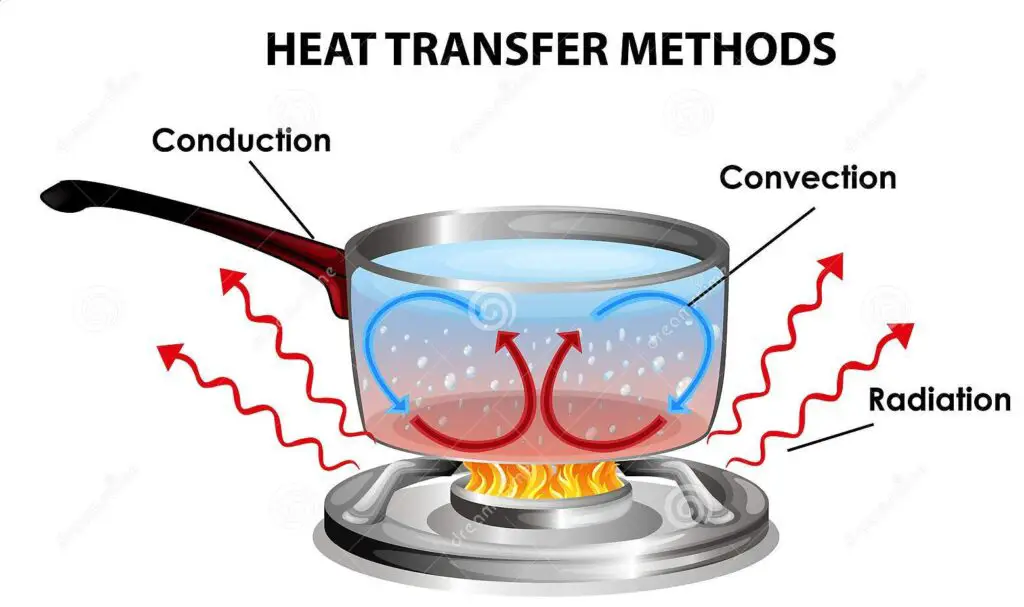
Modes of heat transfer:
Modes are the way how heat is transfer from one body to another body from a object.
There are three modes of:
- Conduction
- Convection
- Radiation
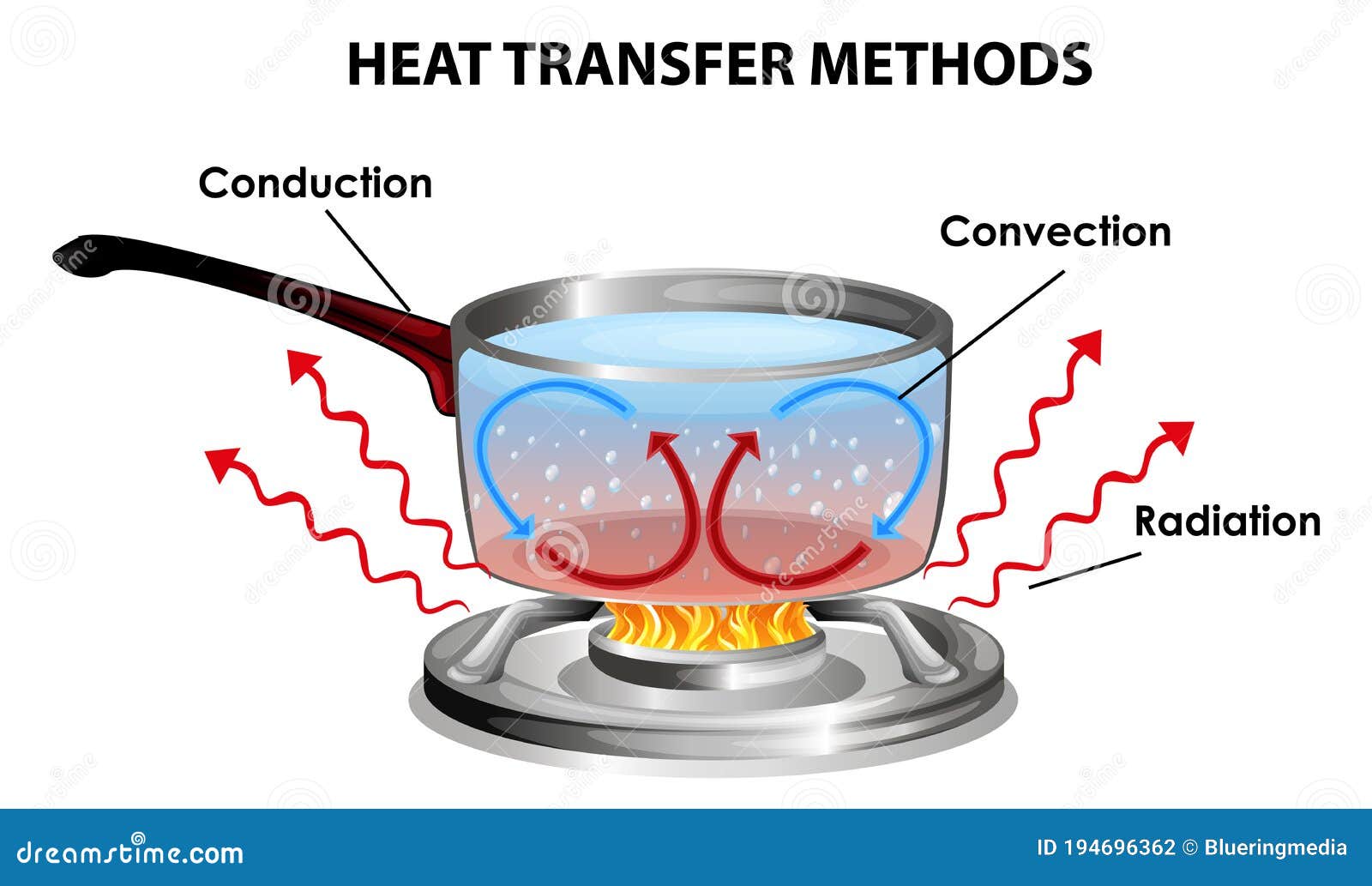
Conduction Heat Transfer:
If the heat is transfer between material by the motion of electron and by the vibration in the lattices molecules is called conduction.
This phenomena generally occurs in the solid where molecules are resisted to flow.
It is also occurs in liquid and gases but effect of conduction in those state are negligible.
Example-
If a steel bar is heated at one end, its temperature rise at other end is an example of heat is transfer by conduction.
LAW OF CONDUCTION / FOURIER LAW OF CONDUCTION-
Fourier law state that-
- Rate of heat transfer is proportional to the temperature gradient exist.
- Rate of heat transfer is proportional to the cross section area of material.
Q = -KA(dX/dt)
(NOTE-Here K is thermal conductivity, Q is heat transfer rate and negative sign is used to fulfill
the condition of second low of thermodynamic.)
Convection:
Convection is the combine effect of heat conduction as well as fluid flow.
In this type of mode heat is conducted or transfer to adjacent layer of fluid by conduction
and then this layer is shifted by any other layer of low temperature through fluid flow.
It is generally occurs in liquid and gases.
Convection depends on the mass flow rate, density of fluid and viscosity of fluid.
LAW OF CONVECTION/ NEWTON’S LAW OF COOLING-
Newton state that heat transfer rate between fluid is –
- Heat transfer rate is proportional to the cross section area.
- Heat transfer rate is proportional to the temperature difference exist.
Q = hA(T2 – T1)
(NOTE- here h is known as convection heat transfer coefficient and Q is heat transfer rate.)
Radiation:
This phenomenon works in vacuum or in absence of a medium by emits the electromagnetic radiation.
Example-
By virtue of this mode of heat transfer sun can able to increase the temperature of earth.
LAW OF RADIATION/ STEFAN BOLTZMAN LAW –
It states that a body will emit energy continuously until its temperature reaches the temperature of surrounding. This energy is proportional to the forth power of temperature and area of body.
Q = σAT4 (for blackbody)
Q =εσAT4 (for non blackbody)
[NOTE – here ε is emissivity, Q is heat transfer rate and σ is Boltzmann constant (σ = 5.669*10-8)]
This is general introduction about heat transfer. We will study every portion of this in detail in later articles. Thanks for reading it.